A Student Balances the Following Redox Reaction Using Half-reactions.
Which is an important step in the alternate method for balancing equations in redox reactions. Balance the two half-reactions separately.

Solved Part B Identifying Redox Reactions Write The Balanced Oxidation And Reduction Half Reactions For Each Redox Reaction A Clz Aq Hbr Aq 7 Hci Aq Brz Aq 6 Sbzoz Aq Kios Aq Hcl Aq Hzo
Consider the redox reaction below.

. Share with your friends. Split the skeletal equation into two half-reactions- oxidation half-reaction and reduction half-reaction. A solution of Iron II nitrate is added to a basic solution of hydrogen peroxide.
Equation 1 HOCl aq I aq H aq _ b. Balancing Redox Equations Using Half-Reactions An overall redox equation can be obtained by. BH4 aq CIO3 aq H2BO3 aq Cl basic b.
Balancing each half 3. Upper A l plus upper M n superscript 2 plus right arrow upper A l superscript 3 plus plus upper M n. Correct answer to the question A student balances the following redox reaction using half-reactions.
Balance the following redox reaction in basic solution. He determines the two half reactions as shown below. Balance the following oxidation-reduction redox reactions using the half reactions method.
Balance the following oxidation-reduction redox reactions using the half reactions method. I- 3H 2 O. Click hereto get an answer to your question Balance the following redox reactions by the ion - electron method in basis medium MnO-4aq I-aq MnO2s I2s.
NO3- Cus à NOg Cu2 b. Balance the following Redox reactions using the half reaction method. Balance the redox reaction below using the half-reaction method.
As noted previously charge balance occurs after mass balance and only uses electrons. How many electrons will be lost in all. VO 2 MnO 4----- VOH 4 Mn 2.
2 Write the balanced reduction half reaction. Equation 2 S2O32 aq I2 aq _ 2. Balance the redox reaction using half reaction method MnO 4-I-MnO 2 IO 3 - in basic medium.
In using the half-reaction method assume that the reaction occurs in an aqueous acid solution Fe2O3 CO Fe CO2 acid solution. Balance the following redox equation using either the oxidation-number-change method or the half-reaction method. S2O32- IO3- àSO42- I-2.
How many electrons will be lost in all. Basic solutions of potassium permanganate and sodium sulfite are added. Zn s NO3- aq Zn2 aq NH3 aq Enter your chemical notation here Balance the following redox reaction in acidic solution using the half reactions method.
Though the oxidation half-reaction was written first in Figure 2 this is not necessary. The balanced reaction is given as. Adding the two half reactions together once the number of e- lost in oxidation are balanced by the e- gained by reduction ex.
Lanay369 lanay369 09242019 Chemistry High School answered A student balances the following redox reaction using half-reactions. NO3- Cu s àNO g Cu2b. On a piece of paper balance and then add the following half-reactions to yield an.
Balance the following redox reactions using the half-reaction method. Use the smallest integer coefficients possible and show electrons as e. S2O32- IO3- Ã SO42- I-2.
Chemistry questions and answers. 3 Write the balanced oxidation half reaction. Balance the following redox reaction.
A student balances the following redox reaction using half-reactions. You must show all your work especially the total number of electrons transferred in the reaction. Breaking into separate reduction and oxidation half-reactions 2.
1 Assign oxidation number to all elements. Get the answers you need now. 4 points Balance the following redox reactions using the half-reaction method AND identify the oxidizing and.
3 on a question A student balances the following redox reaction using half-reactions. Balance the following redox reaction in basic solution using the half reactions method. Balance the following redox reaction in basic solution.
CrO2 aq N2O g Cr3 aq NO g acidic Question. Since the process of oxidation-reduction involves the addition and removal of atoms and charges writing a balanced chemical equation for such reactions is quite a daunting task. Upper A l plus upper M n superscript 2 plus right arrow upper A l superscript 3 plus plus upper M n.
4 points Balance the following redox reactions using the half-reaction method AND identify the oxidizing and reducing agents. MnO 4- I - --- Mn 2 I 2 H 2 O Oxidation half is. But thanks to the redox reaction calculator which makes it easier for students and researchers to balance a complicated redox equation in just a second.
Redox calculator is an online tool which you can. Ir3 aq Hg 1Ir s Hg2² aq a To show your method write the balanced half reactions below. MnO 4 SO 3 2- H 2 O MnO 2 SO 4 2- OH 2 MnO 4 3 SO 3 2- H 2 O 2 MnO 2 3 SO 4 2- 2 OH b.
Ps SO42- Ã PO43- SO32. The two half-reactions are then multiplied by suitable integers so that the total number of electrons gained in the reduction half-reaction is equal to the total number of electrons lost in the. ClO 4- I 2 Cl- IO 3-Balanced.
Coefficients of 1 are not needed. If a box is not needed leave it blank. What is necessary is that the half-reactions be separately balanced both by mass and charge before the final addition.
4 Balance the ionic equation. These are two separate reactions a. Label it using the dipole.
All three reactions are acidic and the ionic species in the reaction are aqueous. CuS s NO3- aq Cu² aq S s NO g Enter your. All three reactions are acidic and the ionic species in the reaction are aqueousa.
Complete and balance the following redox equations by the half reactions method the oxidation reaction and the reduction reaction. A student is asked to balance an equation by using the half-reaction method.

Balancing Redox Equations Br Question Student Doctor Network

Solved Balancing Redox Equations Add The Two Half Reactions Together And Balance The Final Equation By Inspection The Number Of Electrons On Both Sides Must Cancel Oxidation 6fe2t 6fe3t Be Reduction 14h

Redoox Reactions 03 Balancing Redox Reactions In Acidic Solution Youtube

Pin On Science With Tyler Dewitt

Solved Balancing Redox Equations In Acidic Solutions Balance The Following Redox Equations In Acidic Solutions On Separate Paper Write The Final Balanced Equations For The Assigned Redox Reactions On The Oxidation Reduction Equations Dry
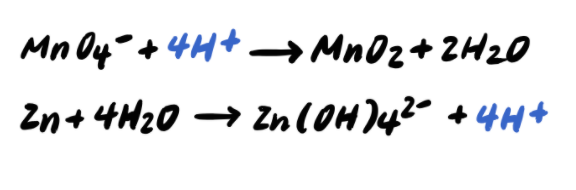
Cc How To Balance Redox Reactions In Acidic And Basic Solutions

Solved Caiculate Ne Cemi Potential Ior Tne Adove Redox Reaction 5 Marks Part D Balancing Redox Reaction Using Oxidation Number 3 Marks Fe O4 S Coia Fets Cozig Cuisl Noz Aql No Cu Nos Zlq Acidic
How To Balance Redox Equations In Basic Solution Schooltube Safe Video Sharing And Management For K12

How To Balance Redox Equations In Basic Solution College Chemistry Equations Chemistry 101

Section 9 2 Balancing Redox Reaction Equations

Redox Reactions Classwork Homework Redox Reactions Covalent Bonding Chemical Equation

Tang 02 Balancing Redox Reactions 2 Redox Reactions Reactions Organic Reactions

Balancing Half Redox Equations Equations Chemistry Positivity

Tang 02 Balancing Redox Reactions 2 Redox Reactions Chemistry Lessons Teaching Chemistry

Solved Balance Redox Reactions Using The Nalf Reaction Method Balance The Redox Reaction Below Using The Half Reaction Method Rh T Aq Pb S Rh S Pb2t Aq To Show You Mnethod Write The Balanced Half Reactions Below Use The
Lesson 2 Balancing Redox Equations Latest Copy Of Grade 12 U

Pin By Saitech Informatics On Balancing Of Chemical Reaction Chemistry Worksheets Equations Redox Reactions

Balancing Redox Equations Half Reactions Basic Solutions Equations Redox Reactions Solutions

Comments
Post a Comment